To legally sell, stock, exhibit, or distribute medical devices and in-vitro diagnostic devices (IVDs) in India, businesses must obtain the required registration under the Medical Devices Rules.
Understanding whether the approval comes from a State Authority or Central Authority is important for compliance.
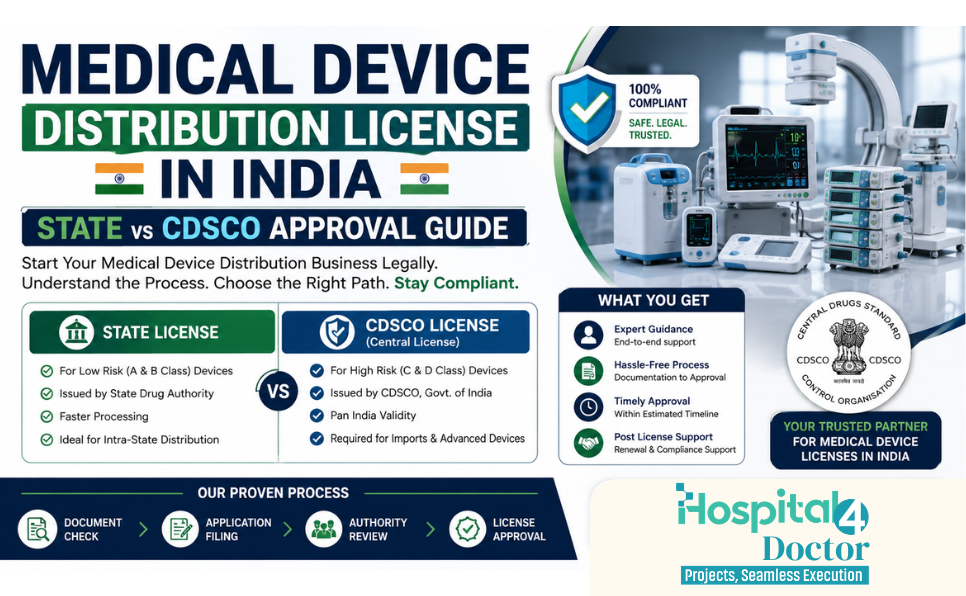
Which License is Required?
✔ Registration Certificate to Sell or Distribute Medical Devices
Application is made in Form MD-41.
This registration is required for:
- Medical device distributors
- Retailers and wholesalers
- Diagnostic equipment suppliers
- IVD product sellers
Which Authority Issues the License?
🏛 State Licensing Authority (State Government)
The registration certificate for sale and distribution of medical devices is generally issued by the:
✔ State Licensing Authority (SLA)
In West Bengal, this falls under the Directorate of Drugs Control, Government of West Bengal.
The State Authority handles:
- Retail & wholesale distribution registration
- Premises verification
- Technical staff compliance
- Local inspections
Where Does the Central Authority Come In?
🏛 Central Drugs Standard Control Organization (CDSCO)
The Central Authority (CDSCO) is involved in:
- Regulation of notified medical devices
- Import licenses
- Manufacturing approvals (certain categories)
- National regulatory standards
CDSCO works under the:
✔ Ministry of Health & Family Welfare, Government of India
State vs Central Authority – Quick Comparison
| Activity | Authority |
|---|---|
| Distribution / Sale License | State Licensing Authority |
| Retail / Wholesale Registration | State Authority |
| Import of Medical Devices | CDSCO (Central) |
| Manufacturing License | State + CDSCO (depending on category) |
| National Regulatory Compliance | CDSCO |
Key Requirements for Registration
Applicants must provide:
✔ Form MD-41 Application
✔ Premises proof
✔ Technical staff qualification
✔ PAN/Aadhaar details
✔ Good Distribution Practice compliance declaration
Why Proper Licensing is Important
Without proper registration:
❌ Sale of medical devices becomes illegal
❌ Risk of penalties and seizure
❌ Regulatory action by authorities
Proper licensing ensures:
✔ Legal business operations
✔ Product traceability
✔ Compliance with Indian medical device laws
How Hospital4Doc Helps
At Hospital4Doc, we provide:
✔ Medical device distribution registration support
✔ State & CDSCO compliance guidance
✔ Documentation & application filing
✔ Technical compliance assistance
✔ End-to-end licensing support
Get Expert Assistance
📞 Call/WhatsApp: +91 98313 55912
🌐 www.hospital4doc.com