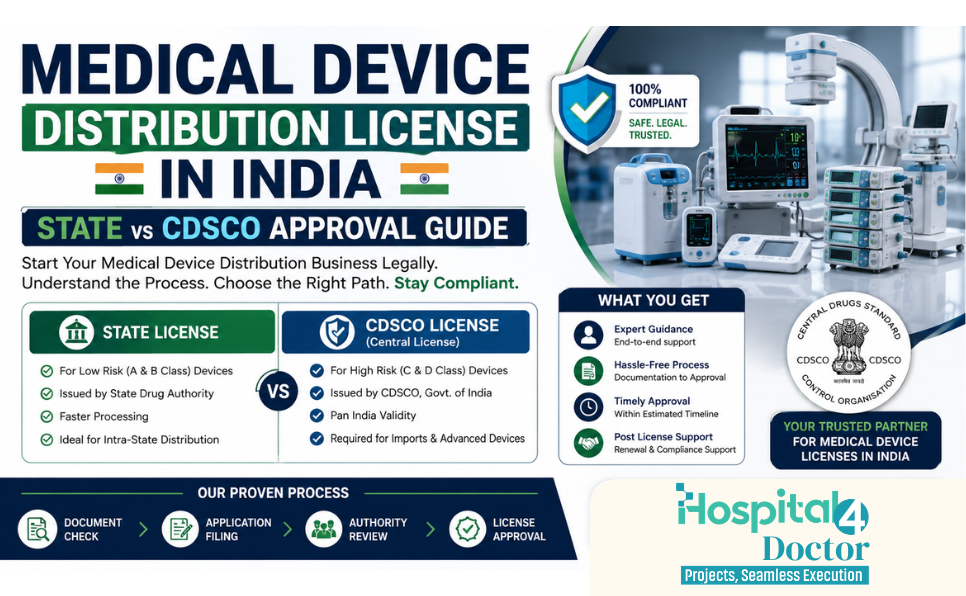
Medical Device Distribution License
To legally sell, stock, exhibit, or distribute medical devices and in-vitro diagnostic devices (IVDs) in India, businesses must obtain the required registration under the Medical Devices Rules. Understanding whether the approval comes from a State Authority or Central Authority is important for compliance. Which License is Required? ✔ Registration Certificate to Sell or Distribute Medical […]
Medical Device Distribution License Read More »